Wellness Chiropractic now offers Erchonia’s FX SERIES Laser for Chronic Low Back Pain of the Musculoskeletal Origin.
FDA market cleared, non-invasive, pain-free procedure with results in 4 weeks
Wellness Chiropractic is now accepting appointments for Erchonia’s new FX SERIES laser treatment for the relief of chronic low back pain of the musculoskeletal origin. The FDA-approved, non-invasive FX SERIES laser is proven to significantly reduce low back pain in just 4 weeks.
Dr. Wehking comments, “Low back pain is extremely painful and persistent and is the leading cause for American’s to miss work. Erchonia’s FX SERIES laser is ideal for low back pain sufferers, because it offers effective, pain-free treatment—without side effects. In just 4 weeks with two treatments per week, patients can have significant and lasting relief from low back pain. Clinical trial results showed an average 58% reduction in pain at 8 weeks post treatment.”
Erchonia’s FX SERIES laser received FDA-clearance based on a double-blind, randomized, multi-site and placebo-controlled clinical trial. Almost all patients treated with the FX SERIES laser noted a dramatic reduction in pain and an improvement in function.
For more information or to book an FX SERIES consultation, please call 920-262-0200.

About Erchonia
Erchonia is the global leader in low level laser healthcare applications. For nearly two decades, Erchonia has been conducting research & development with the world’s leading physicians to advance the science of low level lasers. Erchonia created the low-level laser category after the company was granted the first low level laser FDA clearance for any indication in 2002. Prior to market introduction, all Erchonia lasers are proven to be safe and effective through independent clinical trials. Currently thousands of Erchonia’s lasers are used daily to target body fat and create a slimming effect, target onychomycosis, veterinary applications and reduce pain. For additional information, visit www.erchonia.com.

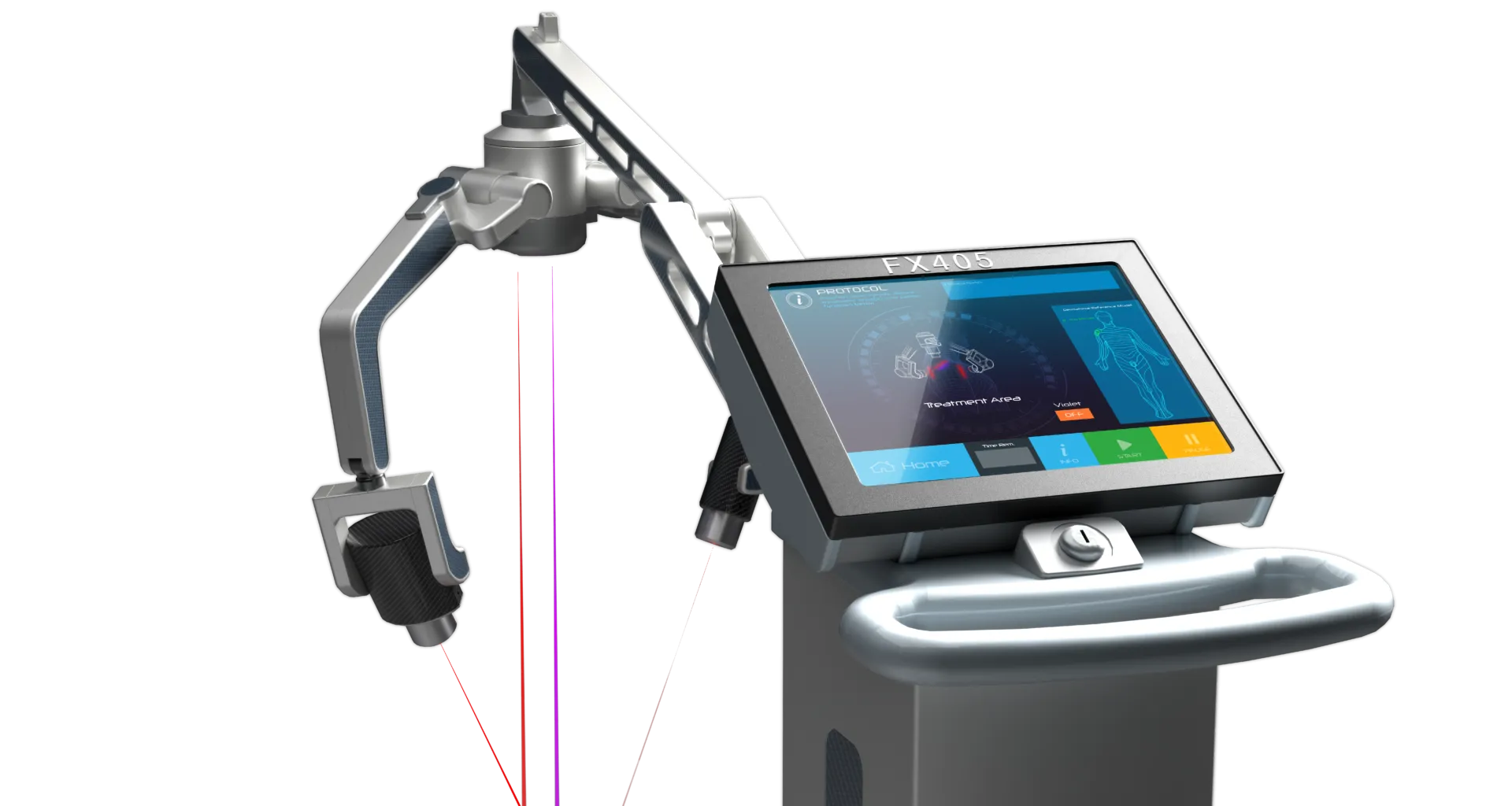
First and Only Chronic Low Back Pain Laser
Discover the only laser on the market for the treatment of chronic low back pain. Our new FDA market cleared laser has been proven in double-blind, placebo-controlled, randomized, parallel group, multi-center clinical trial to effectively treat this painful condition. The participants of the FX 405 Clinical Trial showed a 58% pain reduction at 2 month follow-up evaluation. This trial went on to demonstrate that the FX 405 is successful in the treatment of chronic low back pain. The recent clearance of the FX 405 is groundbreaking for the pain management community and the war on opioids.
How Does FX 405 Work?
The FX 405 uses low-level laser technology to precisely target lower back pain centers. It reduces inflammation while promoting bio-stimulation at a cellular level in the musculoskeletal point of the pain’s origination. Unlike other lasers that are too strong and unreliable — and produce unwanted heat — the FX 405 is fully compliant with ISO 13485 Medical Device Quality and IEC 60825-1 Laser Safety.
Patients with chronic low back pain who choose FX 405 treatment experience improved comfort and quality of life without the risks associated with the side effects of opioids or nonsteroidal anti-inflammatory drugs(NSAIDs) that can cause ulcers, bleeding, kidney disease and heart attacks.

Discover the Fastest Way to Treat Plantar Fasciitis
Now you can relieve the pain and inflammation of the heel associated with plantar fasciitis. Our new FDA market cleared laser has been proven in clinical tests to effectively treat this painful condition. The regenerative powers of this low level laser technology can reduce inflammation and eliminate pain in the plantar fascia in only a matter of weeks and help you return to an active life once again.